Hydrogen-air Combustion CFD Simulation: NOx Formation and Radiation in Fluent
Hydrogen-air Combustion CFD Simulation: NOx Formation and Radiation in Fluent
- Upon ordering this product, you will be provided with a geometry file, a mesh file, and an in-depth Training Video that offers a step-by-step training on the simulation process.
- For any more inquiries regarding the product, please do not hesitate to reach out to us at info@CFDLAND.com or through our online support assistant.
€240 Original price was: €240.€135Current price is: €135.
Hydrogen is becoming a popular clean fuel because it produces mainly water when it burns. However, Hydrogen-air Combustion releases a massive amount of heat. This leads to extremely high temperatures inside the engine or burner. While high temperature is good for efficiency, it creates two big problems that engineers must solve using CFD simulation.
First, the intense heat creates Thermal NOx. This is a harmful pollutant formed when nitrogen in the air reacts with oxygen at high temperatures. Predicting this requires a precise NOx formation simulation. Second, a lot of energy is lost through radiation, which changes the temperature field. Therefore, an accurate Hydrogen combustion CFD model must include both Radiation modeling and NOx prediction. This report presents a detailed CFD study of a micro-cylindrical combustor. We use ANSYS Fluent to model the fluid flow, heat transfer, and chemical reactions. For more guides on reacting flows, please visit our combustion tutorials. The simulation methodology is guided by the physical principles discussed in the study by Pashchenko [1].
- Reference [1]: Pashchenko, Dmitry. “Comparative analysis of hydrogen/air combustion CFD-modeling for 3D and 2D computational domain of micro-cylindrical combustor.” International Journal of Hydrogen Energy49 (2017): 29545-29556.
- Reference [2]: Jiaqiang, E., et al. “Effects of inlet pressure on wall temperature and exergy efficiency of the micro-cylindrical combustor with a step.” Applied Energy175 (2016): 337-345.
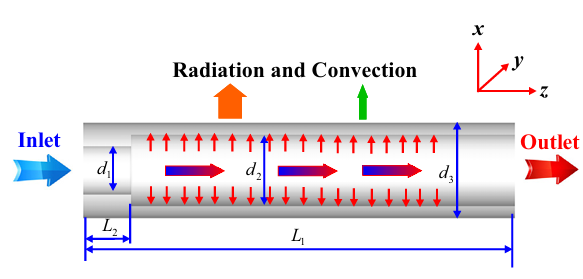
Figure 1: Schematic diagram of the micro-cylindrical combustor design. [2].
Simulation Process: 3D Combustion CFD Modeling in ANSYS Fluent
The simulation process started with building a precise 3D model in ANSYS Design Modeler. The geometry of the micro-combustor is complex, consisting of 26 separate bodies. This multi-body approach was necessary to create a high-quality structured mesh. In ANSYS Meshing, we generated a grid with 164,160 cells. A structured mesh is crucial for Combustion CFD because it aligns with the flow direction, reducing numerical diffusion and increasing the accuracy of the chemical reaction calculations.
The physics setup in ANSYS Fluent was comprehensive. We enabled the Species Transport model to handle the mixing and burning of hydrogen and air. Instead of a simple one-step reaction, we used a detailed 19-step reaction mechanism. This mechanism tracks many intermediate species, which is essential for accurate NOx formation combustion analysis. To capture the heat loss, we activated the P1 Radiation model. Finally, we turned on the NOx model to calculate the formation of pollutants based on the temperature and radical concentrations. This setup creates a robust Hydrogen flame simulation capable of predicting real-world phenomena.

Figure 2: The 3D geometry of the combustion chamber showing the multi-body structure.

Figure 3: The high-quality structured mesh used for the CFD analysis.
Post-processing: Analysis of Flame Structure and Pollutants
In this CFD study, we analyze the internal structure of the flame to understand how pollutants form. Figure 4 shows the mass fraction of Hydrogen Combustion Products. The H2O (Water Vapor) contour shows where the combustion is complete. The concentration is highest at the outlet, indicating that the hydrogen has successfully burned. More importantly, we look at the Atomic Oxygen (O) contour. Atomic Oxygen is a highly reactive radical. It only exists in the hottest parts of the flame where oxygen molecules break apart. Its presence is a marker for the reaction zone. In a NOx formation simulation, knowing the location of Atomic Oxygen is vital because it is a key player in the Zeldovich mechanism (Thermal NOx). The high concentration of ‘O’ tells us exactly where the temperature is high enough to trigger NOx production.
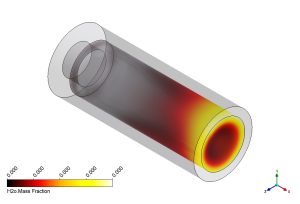
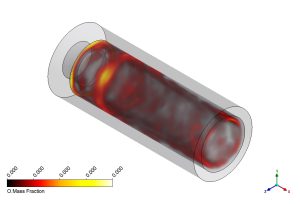
Figure 4: Mass fraction contours of H2O and Atomic O radicals.
Figure 5 visualizes the flow path using streamlines colored by Hydrogen Peroxide (H2O2). H2O2 is another intermediate species tracked by our 19-step mechanism. The smooth streamlines show that the flow moves steadily through the chamber. The presence of H2O2 indicates the early stages of ignition and low-temperature oxidation regions. This level of detail confirms that the Species Transport model is working correctly. By accurately predicting these intermediate species and the temperature field (thanks to the P1 Radiation model), ANSYS Fluent can calculate the NOx emission rates with high confidence. This study demonstrates that CFD is a powerful tool for designing cleaner hydrogen combustors without needing expensive physical testing.
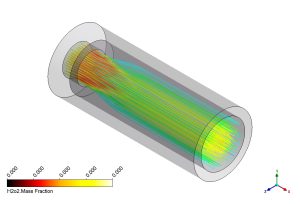
Figure 5: Streamlines colored by H2O2 mass fraction showing the flow path.
Key Takeaways & FAQ
- Q: Why does Hydrogen combustion produce NOx if there is no Carbon?
- A: Although Hydrogen (H2) has no carbon, the air used to burn it contains 78% Nitrogen (N2). At the very high temperatures of Hydrogen-air combustion, the nitrogen and oxygen in the air react with each other to form Nitrogen Oxides (NOx). This is known as Thermal NOx.
- Q: Why is the P1 Radiation model important in this simulation?
- A: Hydrogen flames are very hot. A significant amount of this heat is released as radiation. If we ignore radiation in the CFD simulation, the predicted temperatures will be too high. Since NOx formation is extremely sensitive to temperature (exponential dependence), incorrect temperatures would lead to wrong NOx predictions.
- Q: What is the benefit of a 19-step reaction mechanism?
- A: A simple “Fuel + Oxidizer -> Product” reaction cannot capture the complex physics of combustion. A 19-step mechanism tracks intermediate radicals like O, H, and OH. These radicals are critical for predicting flame stability and represent the actual chemical pathways that lead to NOx formation.
We pride ourselves on presenting unique products at CFDLAND. We stand out for our scientific rigor and validity. Our products are not based on guesswork or theoretical assumptions like many others. Instead, most of our products are validated using experimental or numerical data from valued scientific journals. Even if direct validation isn’t possible, we build our models and assumptions on the latest research, typically using reference articles to approximate reality.
Yes, we’ll be here . If you have trouble loading files, having technical problems, or have any questions about how to use our products, our technical support team is here to help.
You can load geometry and mesh files, as well as case and data files, using any version of ANSYS Fluent.
€190 Original price was: €190.€125Current price is: €125.

€240 Original price was: €240.€125Current price is: €125.

€295 Original price was: €295.€175Current price is: €175.

€245 Original price was: €245.€185Current price is: €185.

€360 Original price was: €360.€185Current price is: €185.

€245 Original price was: €245.€199Current price is: €199.




















Reviews
There are no reviews yet.