Hydrogen Addition Effect on NOx Formation CFD Simulation: Combustion Analysis in Fluent
Hydrogen Addition Effect on NOx Formation CFD Simulation: Combustion Analysis in Fluent
- Upon ordering this product, you will be provided with a geometry file, a mesh file, and an in-depth Training Video that offers a step-by-step training on the simulation process.
- For any more inquiries regarding the product, please do not hesitate to reach out to us at info@CFDLAND.com or through our online support assistant.
€125
Hydrogen (H2) is often called the fuel of the future because it contains no carbon, meaning it produces zero Carbon Dioxide (CO2) when burned. However, burning hydrogen presents a different challenge for engineers: NOx Formation. Nitrogen Oxides (NOx) are harmful pollutants that form when nitrogen and oxygen in the air react at very high temperatures. Because hydrogen burns much hotter and faster than natural gas (Methane), adding it to fuel blends can drastically increase these emissions.
This project is a Hydrogen Addition Effect on NOx Formation CFD simulation designed to analyze this specific problem. We will use ANSYS Fluent to visualize the chemical reactions and temperature fields that are impossible to see with the naked eye. We will investigate how adding small amounts of hydrogen changes the flame structure and pollution output. For more in-depth learning on reacting flows, please visit our Combustion tutorials. Our simulation setup is based on the parameters found in the research by Ali et al. [1].
- Reference [1]: Ali, Ghufran, et al. “Effect of hydrogen addition on NOx formation mechanism and pathways in MILD combustion of H2-rich low calorific value fuels.” International Journal of Hydrogen Energy15 (2020): 9200-9210.
Simulation process: Modeling Species Transport in Fluent
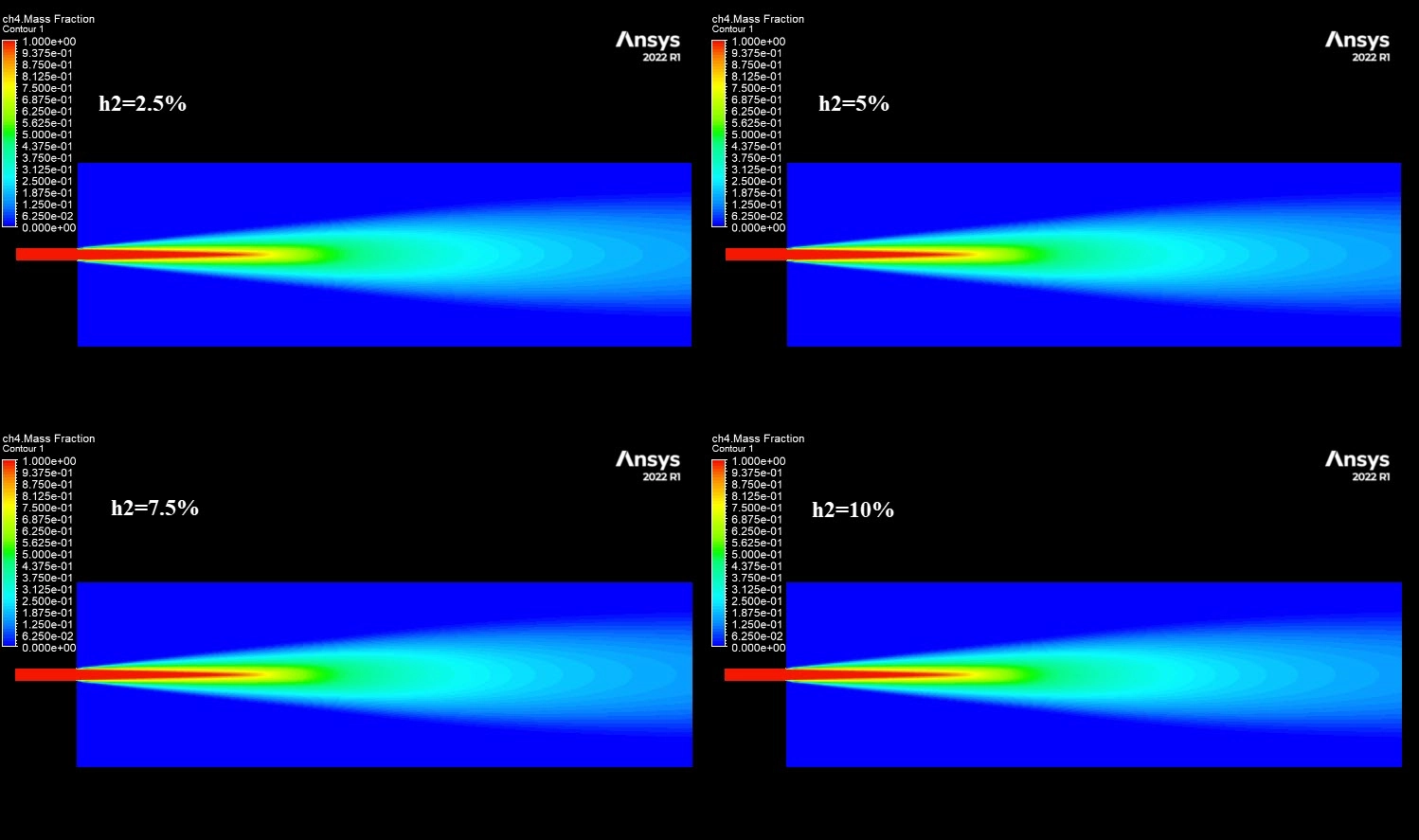
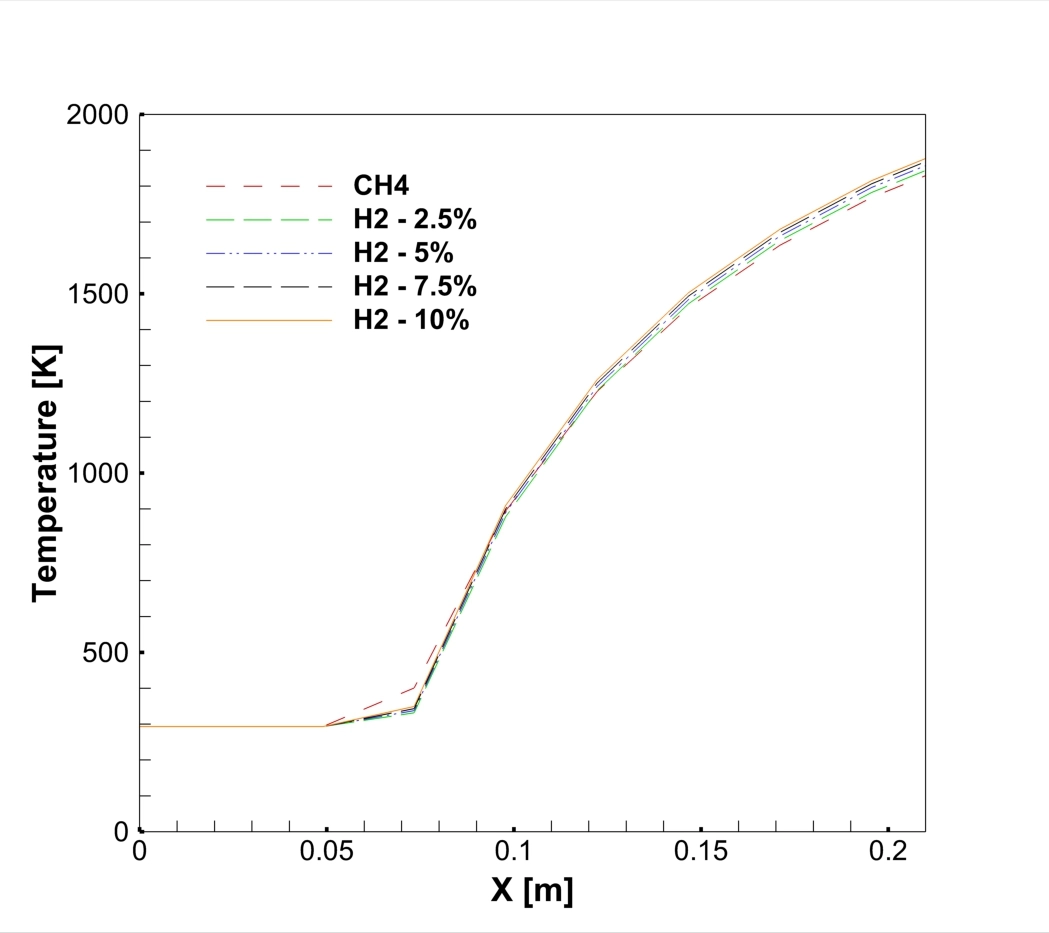
To perform this Hydrogen Addition fluent simulation, we started by creating a 2D axisymmetric geometry representing a cylindrical burner. We generated a structured mesh with rectangular cells, refining the grid near the fuel inlet to capture the mixing zone accurately. In ANSYS Fluent, we selected the Species Transport model. This model is essential because it calculates the mixing and reaction of the different chemical species: Methane (CH4), Hydrogen (H2), Oxygen (O2), and Nitrogen (N2).
The core of this Hydrogen Addition ANSYS Fluent study lies in the NOx modeling. We activated the NOx model within the solver, specifically selecting the Thermal NOx and Prompt NOx pathways. Thermal NOx, governed by the Zeldovich mechanism, is the most critical factor here because it depends entirely on temperature. We set up four different cases with varying hydrogen content: 0% (pure Methane), 2.5%, 5%, 7.5%, and 10%. By keeping the total energy input constant but changing the fuel composition, we can isolate the exact Hydrogen Addition Effect on NOx Formation Simulation.
Post-processing: Analysis of Thermal NOx and Flame Speed
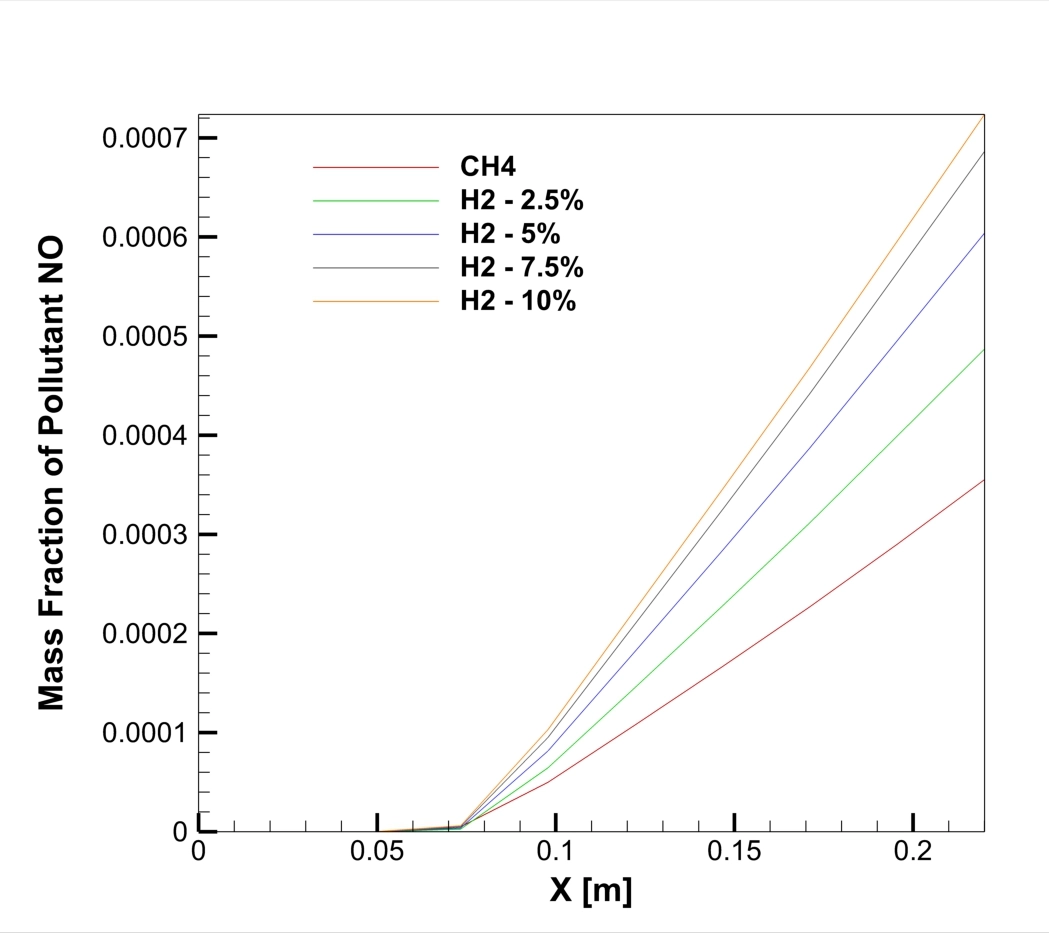
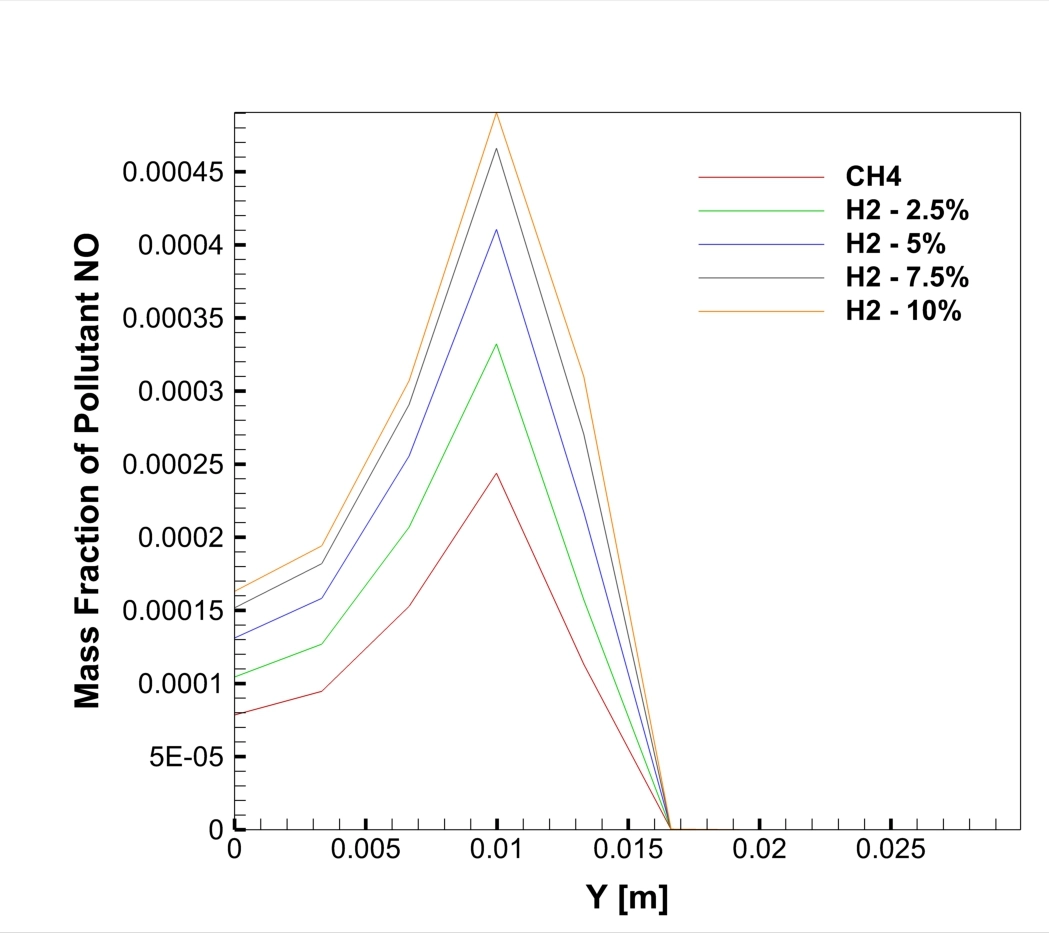
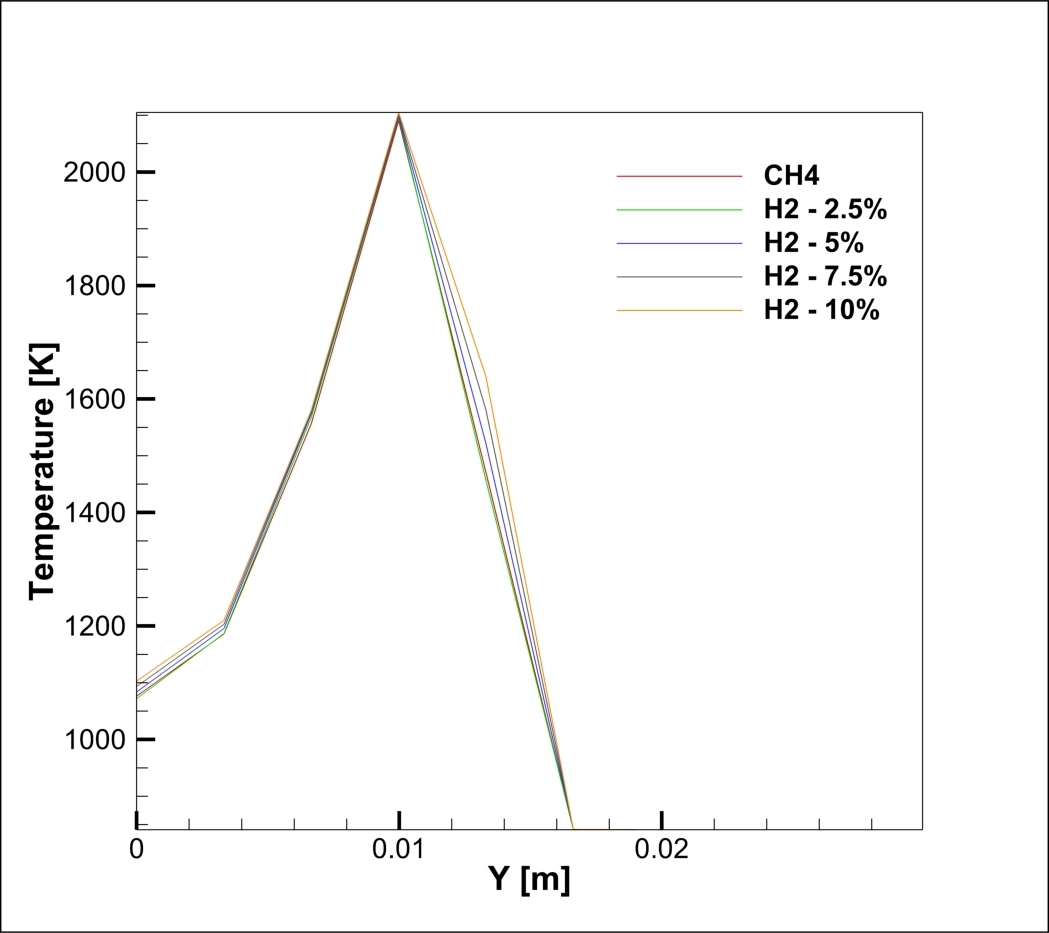
A real analysis of the simulation results, based on the specific data and contours provided, reveals a direct linear relationship between hydrogen content and pollution. The graph of NO Mass Fraction at the outlet (Figure 1) tells the main story. The red line, representing pure Methane (0% H2), shows a peak NOx concentration of roughly 240 ppm (0.00024 mass fraction). However, as we look at the orange line representing the 10% Hydrogen case, the peak concentration jumps to 480 ppm (0.00048 mass fraction). This is a massive increase; adding just 10% hydrogen effectively doubles the NOx emissions. The graph also shows that for every 2.5% of hydrogen added, the emission levels rise by approximately 50 to 60 ppm. The peak location at the mid-radius (Y = 0.01 m) identifies the “flame front,” the hottest part of the combustion zone where fuel and oxidizer meet.
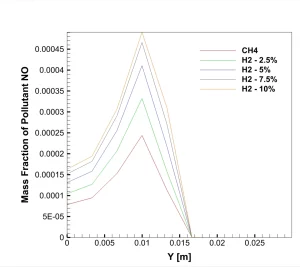
Figure 1: The NO Mass Fraction graph showing the increase in emissions as Hydrogen percentage rises.
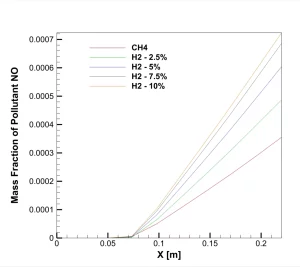
Figure 2: NO mass fraction on centerline
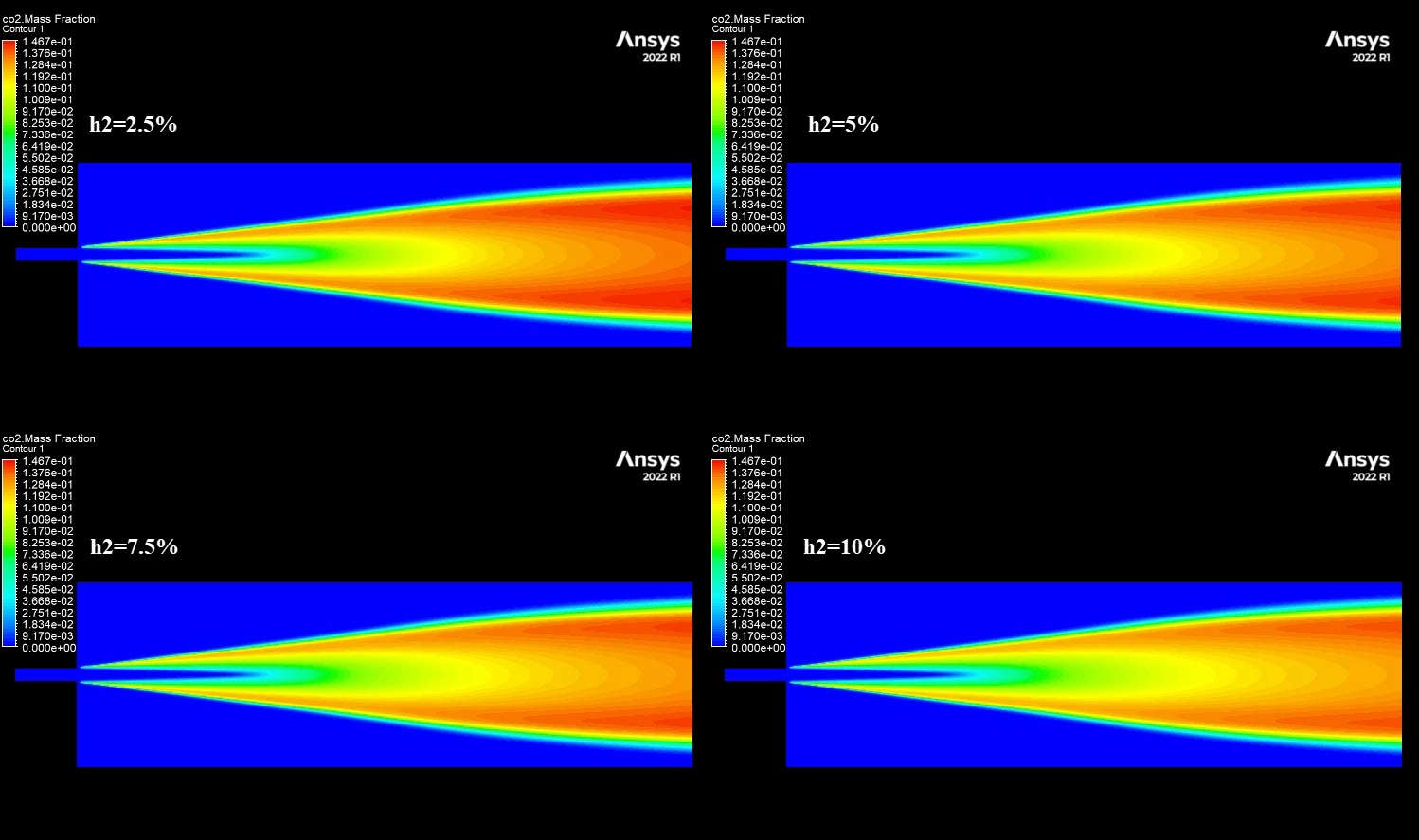
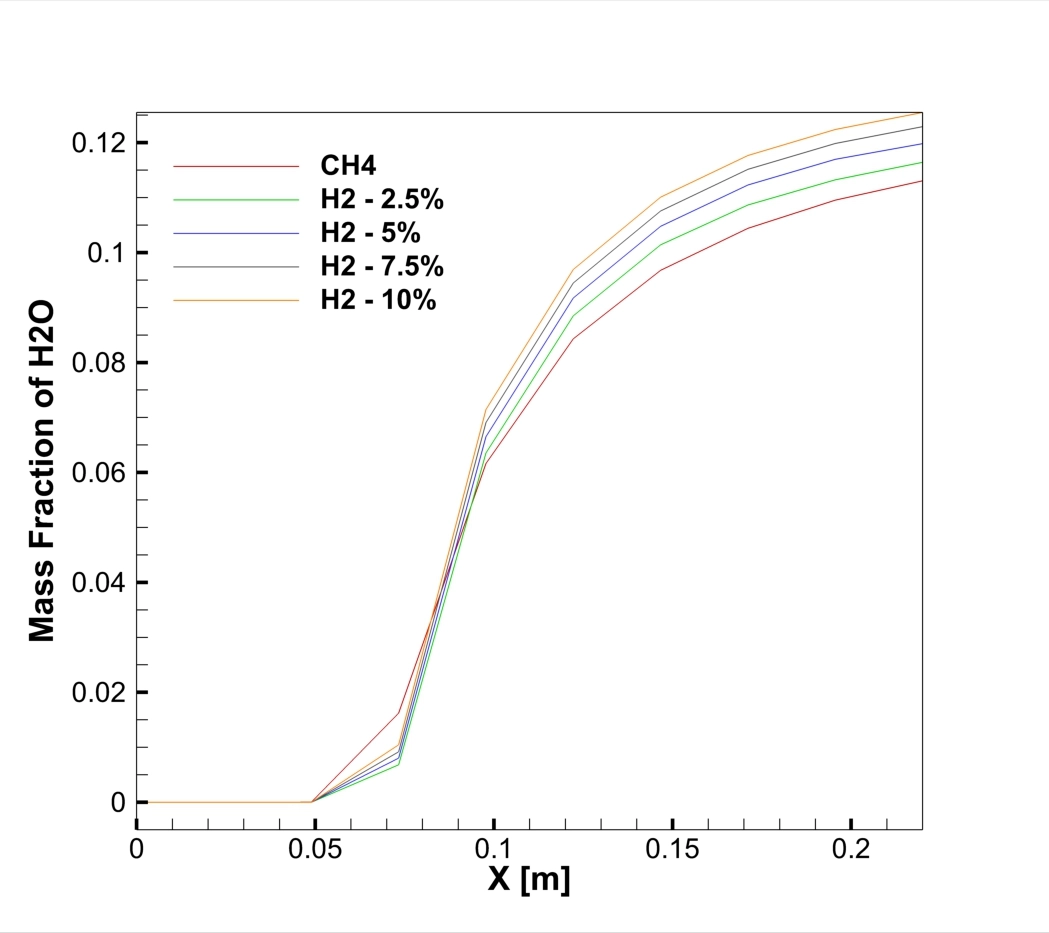
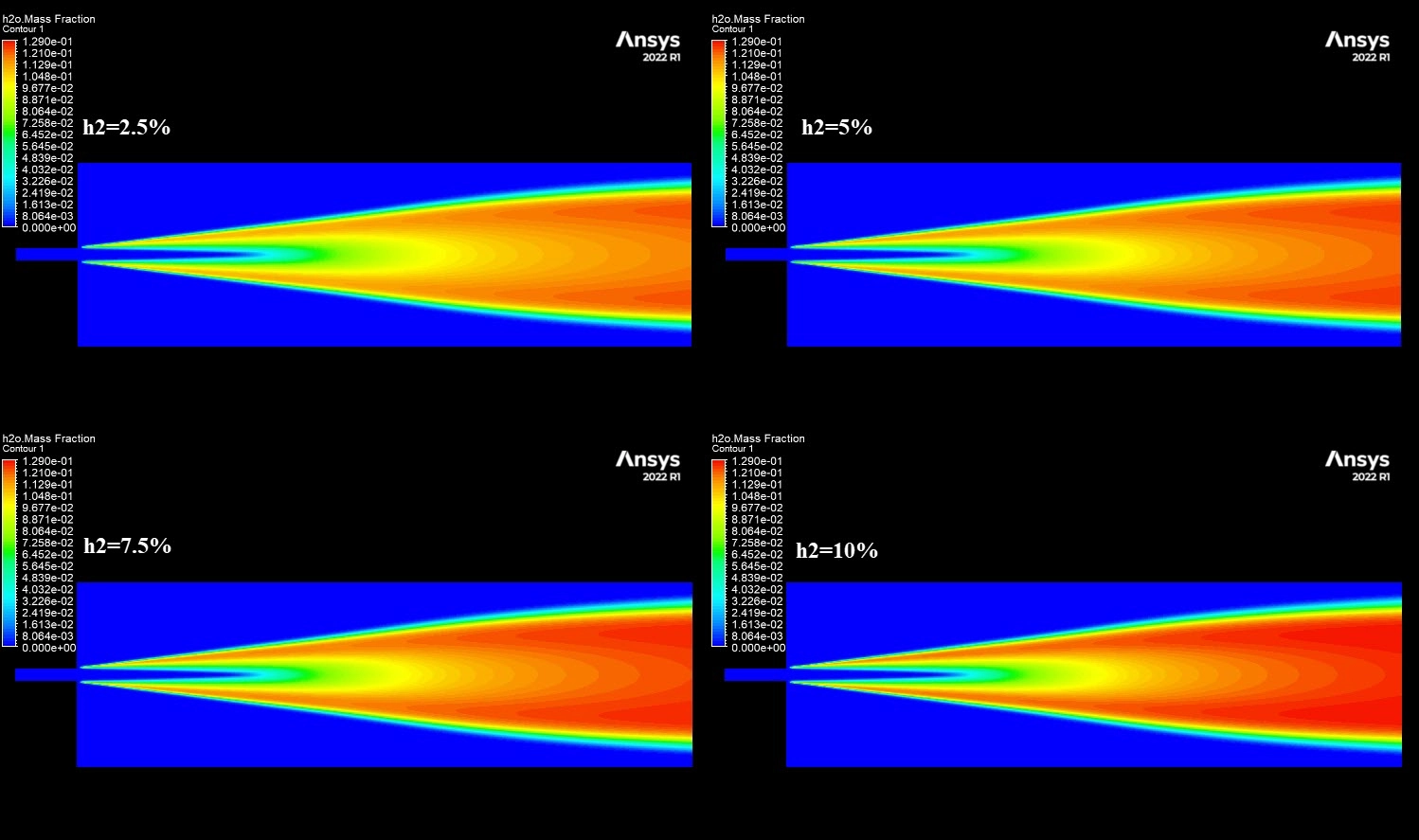
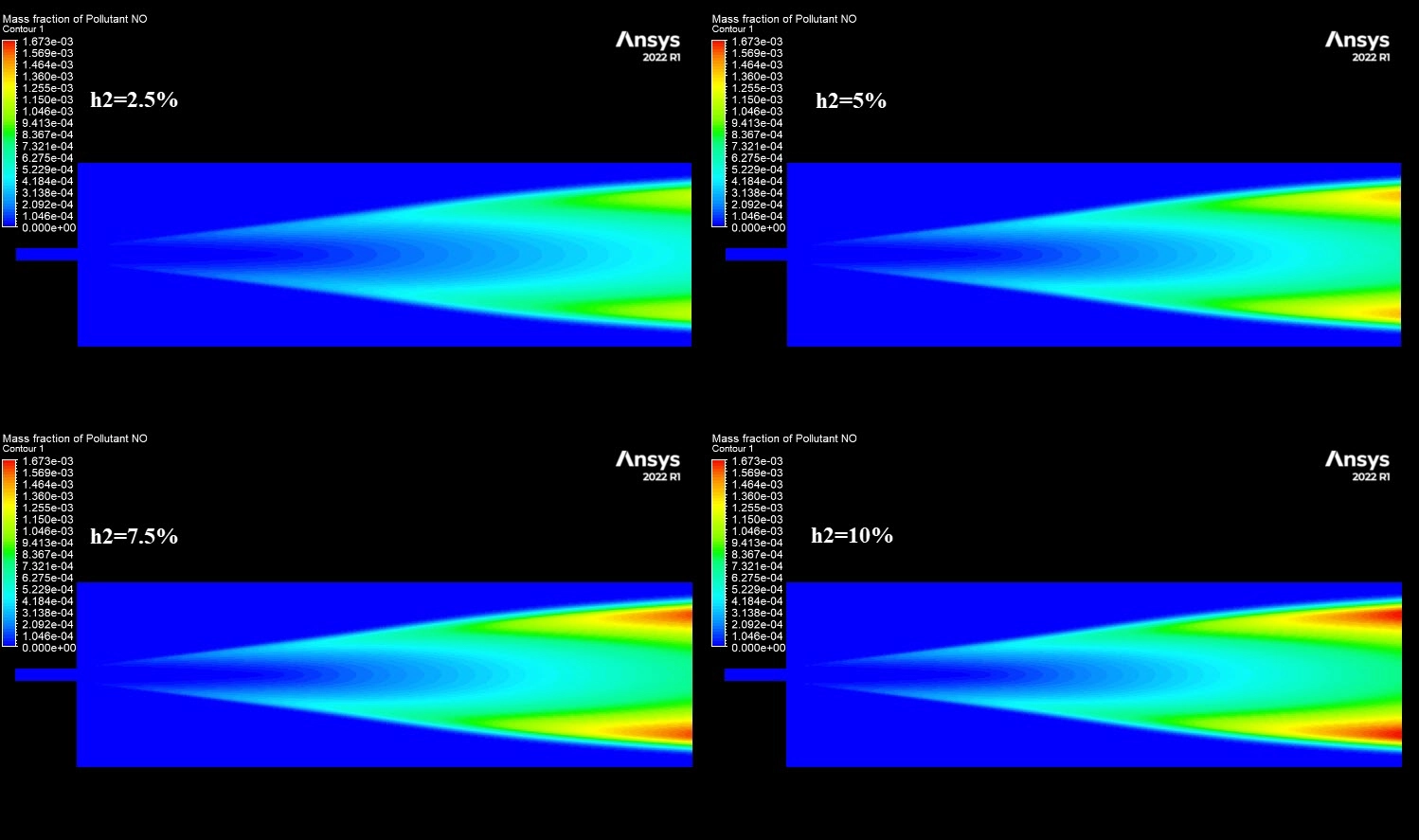
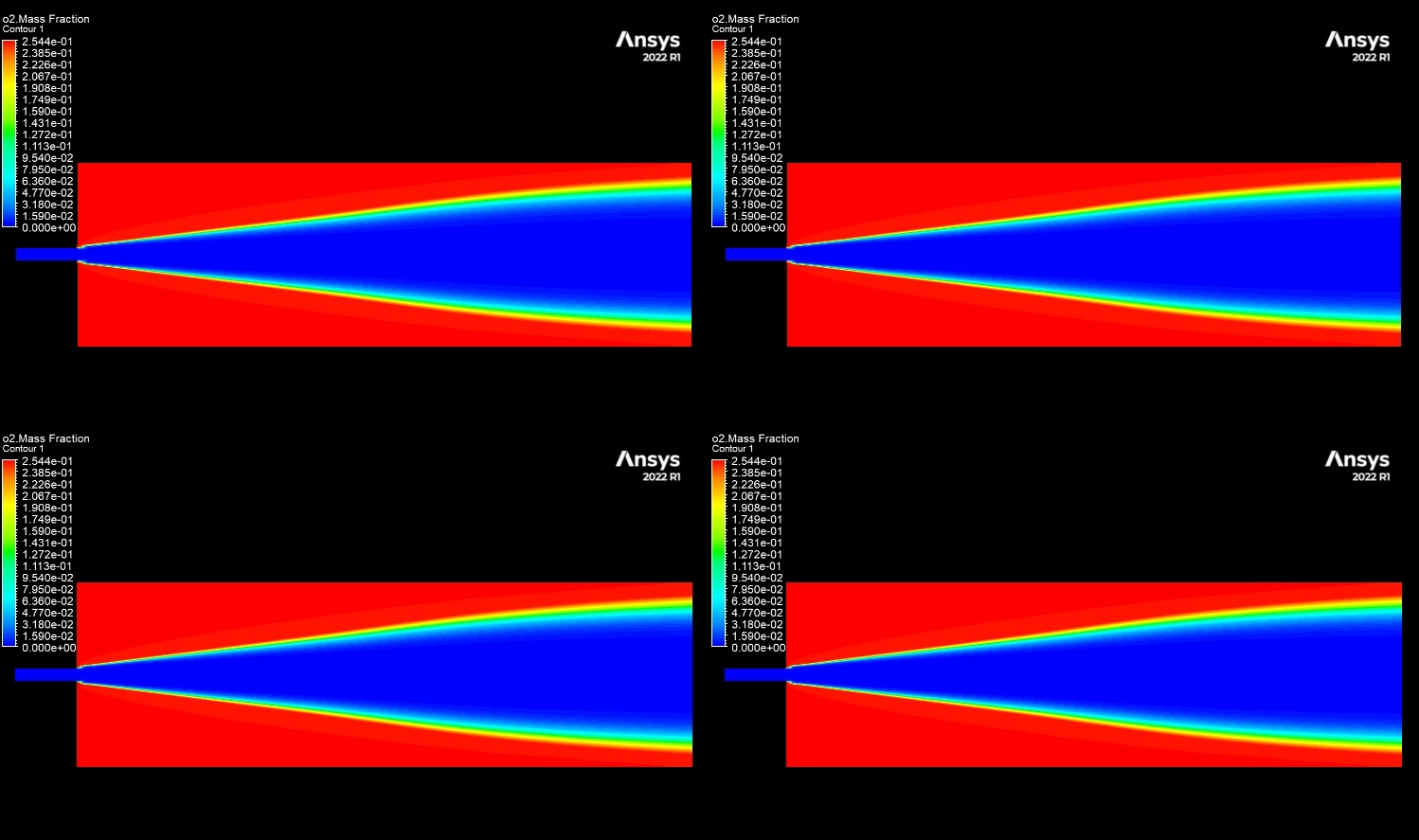
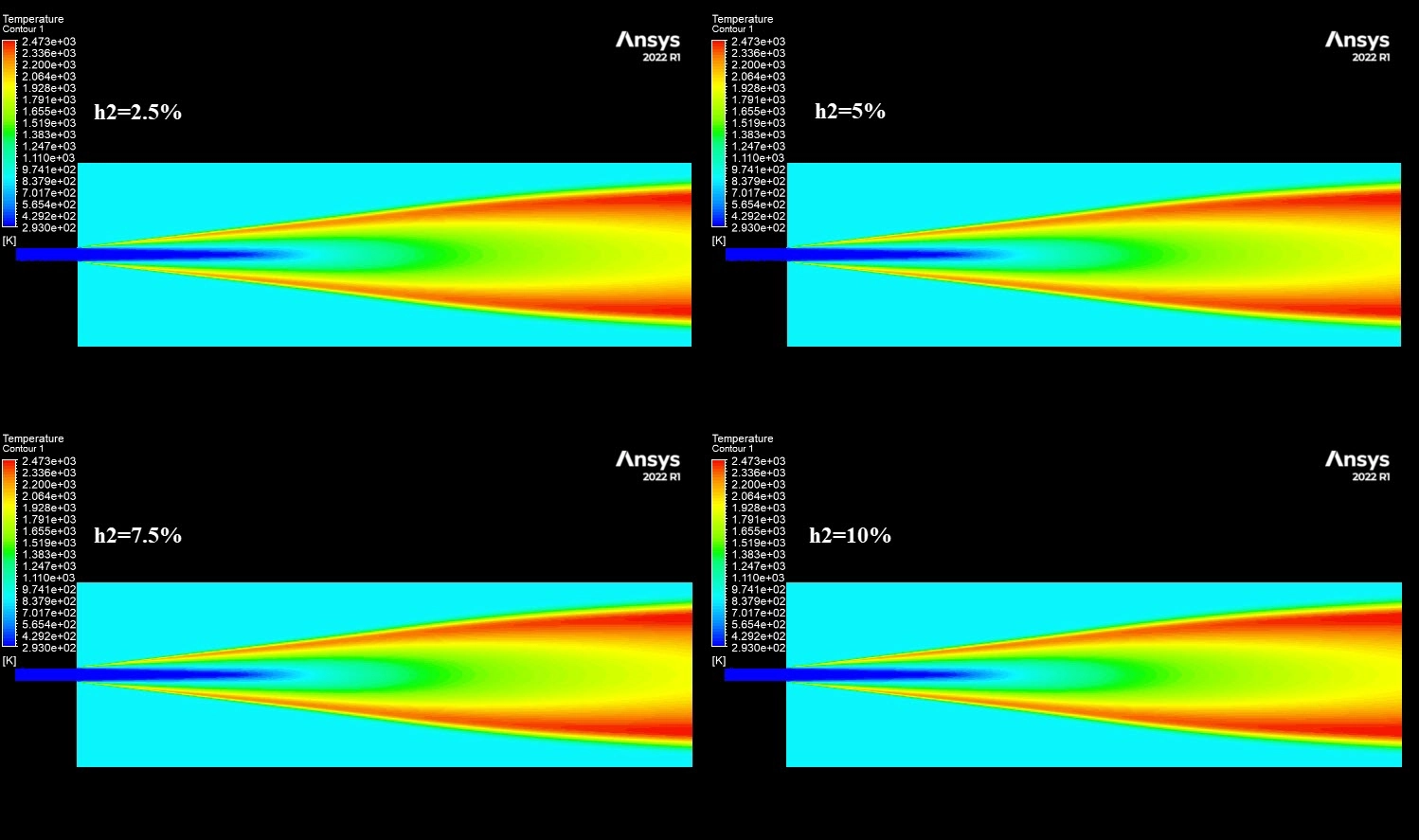
The contours in Figure 3 visually explain the physics behind these numbers. The 10% H2 case is dominated by large red zones, indicating very high local NOx concentrations reaching a maximum of 1673 ppm. The primary driver for this is the burning velocity. Hydrogen has a laminar flame speed of 2-3 m/s, which is significantly faster than Methane’s 0.4 m/s. This rapid combustion releases heat intensely and instantaneously, creating localized hot spots. Since the Zeldovich mechanism for Thermal NOx is exponentially sensitive to temperature (the reaction rate doubles for every 90 K increase), these hotter flames trigger massive NOx production. Furthermore, while the H2O contours show that hydrogen produces more water vapor (up to 0.129 mass fraction) which typically cools a flame, the sheer Heating Value of hydrogen (120 MJ/kg) overpowers this cooling effect. This CFD Analysis of Hydrogen Addition Effect on Nox Formation conclusively proves that while hydrogen reduces carbon emissions, it requires careful thermal management to prevent a spike in harmful nitrogen oxides.
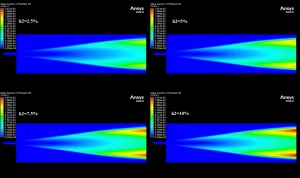
Figure 3: NO mass fraction contours comparing low and high hydrogen cases.
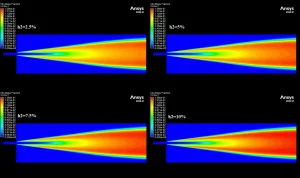
Figure 4: H2O mass fraction contours showing the water vapor core.
Key Takeaways & FAQ
- Q: Why does hydrogen addition increase NOx formation?
- A: As shown in this Hydrogen Addition Effect on NOx Formation CFD simulation, hydrogen burns much faster (2-3 m/s) and hotter than methane. This high temperature triggers the Zeldovich mechanism, creating large amounts of Thermal NOx.
- Q: What is the Zeldovich mechanism?
- A: The Zeldovich mechanism is a chemical pathway where atmospheric nitrogen (N2) reacts with oxygen (O2) at high temperatures (usually above 1300°C) to form Nitric Oxide (NO). It is the primary source of NOx in hydrogen combustion.
- Q: What models are used in ANSYS Fluent for this?
- A: We use the Species Transport model to handle the combustion chemistry and the dedicated NOx model (selecting Thermal and Prompt pathways) to predict the pollutant formation concentrations.
We pride ourselves on presenting unique products at CFDLAND. We stand out for our scientific rigor and validity. Our products are not based on guesswork or theoretical assumptions like many others. Instead, most of our products are validated using experimental or numerical data from valued scientific journals. Even if direct validation isn’t possible, we build our models and assumptions on the latest research, typically using reference articles to approximate reality.
Yes, we’ll be here . If you have trouble loading files, having technical problems, or have any questions about how to use our products, our technical support team is here to help.
You can load geometry and mesh files, as well as case and data files, using any version of ANSYS Fluent.
€135 Original price was: €135.€65Current price is: €65.

€360 Original price was: €360.€185Current price is: €185.

€240 Original price was: €240.€175Current price is: €175.

€180 Original price was: €180.€115Current price is: €115.














Reviews
There are no reviews yet.